Product Description
CRYBB2 Antibody | 19-740 | ProSci
Host: Rabbit
Reactivity: Human, Mouse, Rat
Homology: N/A
Immunogen: Recombinant fusion protein containing a sequence corresponding to amino acids 1-205 of human CRYBB2 (NP_000487.1) .
Research Area: Other
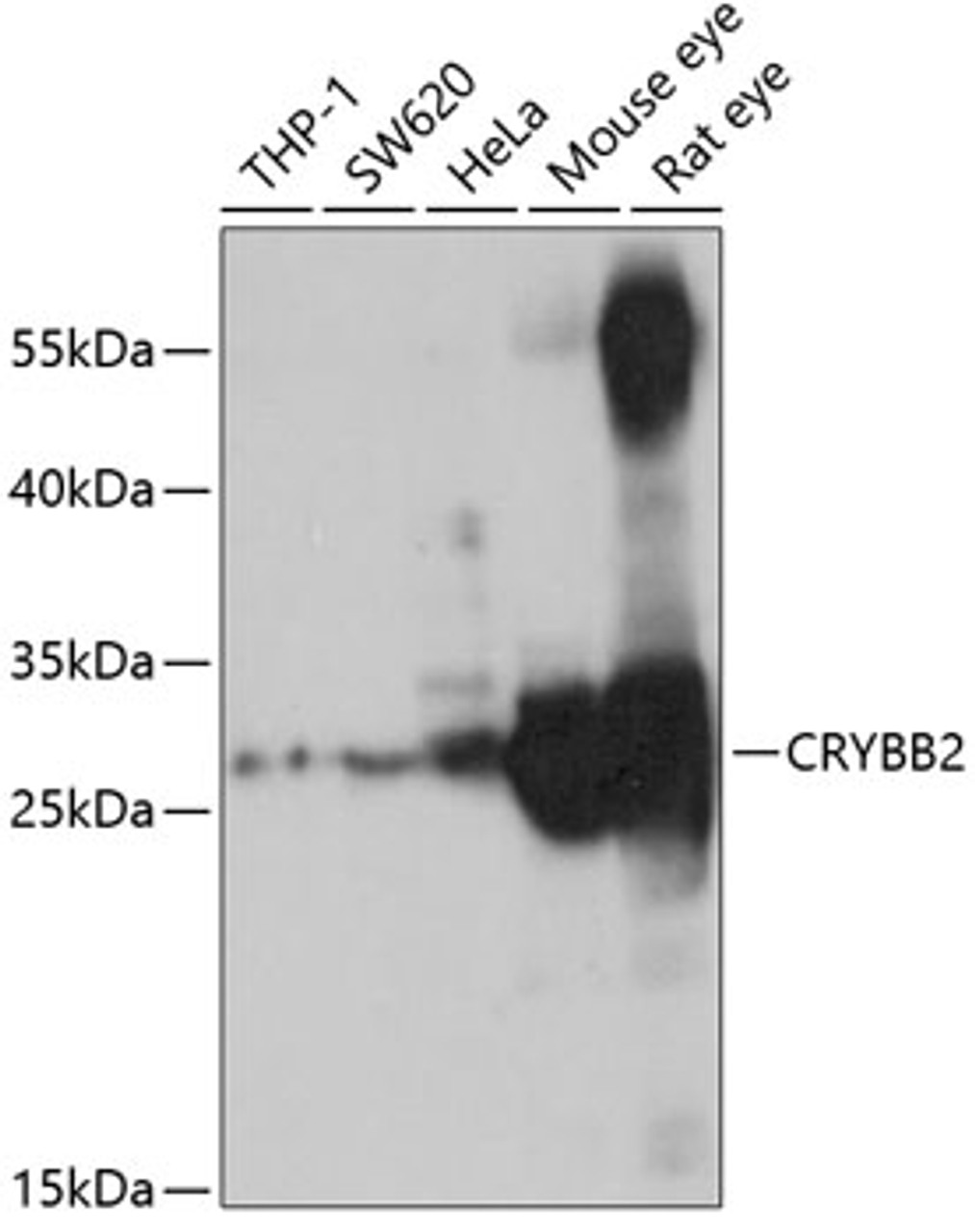
Tested Application: WB
Application: WB: 1:500 - 1:2000
Specificiy: N/A
Positive Control 1: THP-1
Positive Control 2: SW620
Positive Control 3: HeLa
Positive Control 4: Mouse eye
Positive Control 5: Rat eye
Positive Control 6: N/A
Molecular Weight: Observed: 27kDa
Validation: N/A
Isoform: N/A
Purification: Affinity purification
Clonality: Polyclonal
Clone: N/A
Isotype: IgG
Conjugate: Unconjugated
Physical State: Liquid
Buffer: PBS with 0.02% sodium azide, 50% glycerol, pH7.3.
Concentration: N/A
Storage Condition: Store at -20˚C. Avoid freeze / thaw cycles.
Alternate Name: CCA2, CRYB2, CRYB2A, CTRCT3, D22S665, beta-crystallin B2, CTA-221G9.7, beta-B2 crystallin, beta-crystallin Bp, eye lens structural protein
User Note: Optimal dilutions for each application to be determined by the researcher.
BACKGROUND: Crystallins are separated into two classes: taxon-specific, or enzyme, and ubiquitous. The latter class constitutes the major proteins of vertebrate eye lens and maintains the transparency and refractive index of the lens. Since lens central fiber cells lose their nuclei during development, these crystallins are made and then retained throughout life, making them extremely stable proteins. Mammalian lens crystallins are divided into alpha, beta, and gamma families; beta and gamma crystallins are also considered as a superfamily. Alpha and beta families are further divided into acidic and basic groups. Seven protein regions exist in crystallins: four homologous motifs, a connecting peptide, and N- and C-terminal extensions. Beta-crystallins, the most heterogeneous, differ by the presence of the C-terminal extension (present in the basic group, none in the acidic group) . Beta-crystallins form aggregates of different sizes and are able to self-associate to form dimers or to form heterodimers with other beta-crystallins. This gene, a beta basic group member, is part of a gene cluster with beta-A4, beta-B1, and beta-B3. A chain-terminating mutation was found to cause type 2 cerulean cataracts.
 Euro
Euro
 USD
USD
 British Pound
British Pound
 NULL
NULL