Product Description
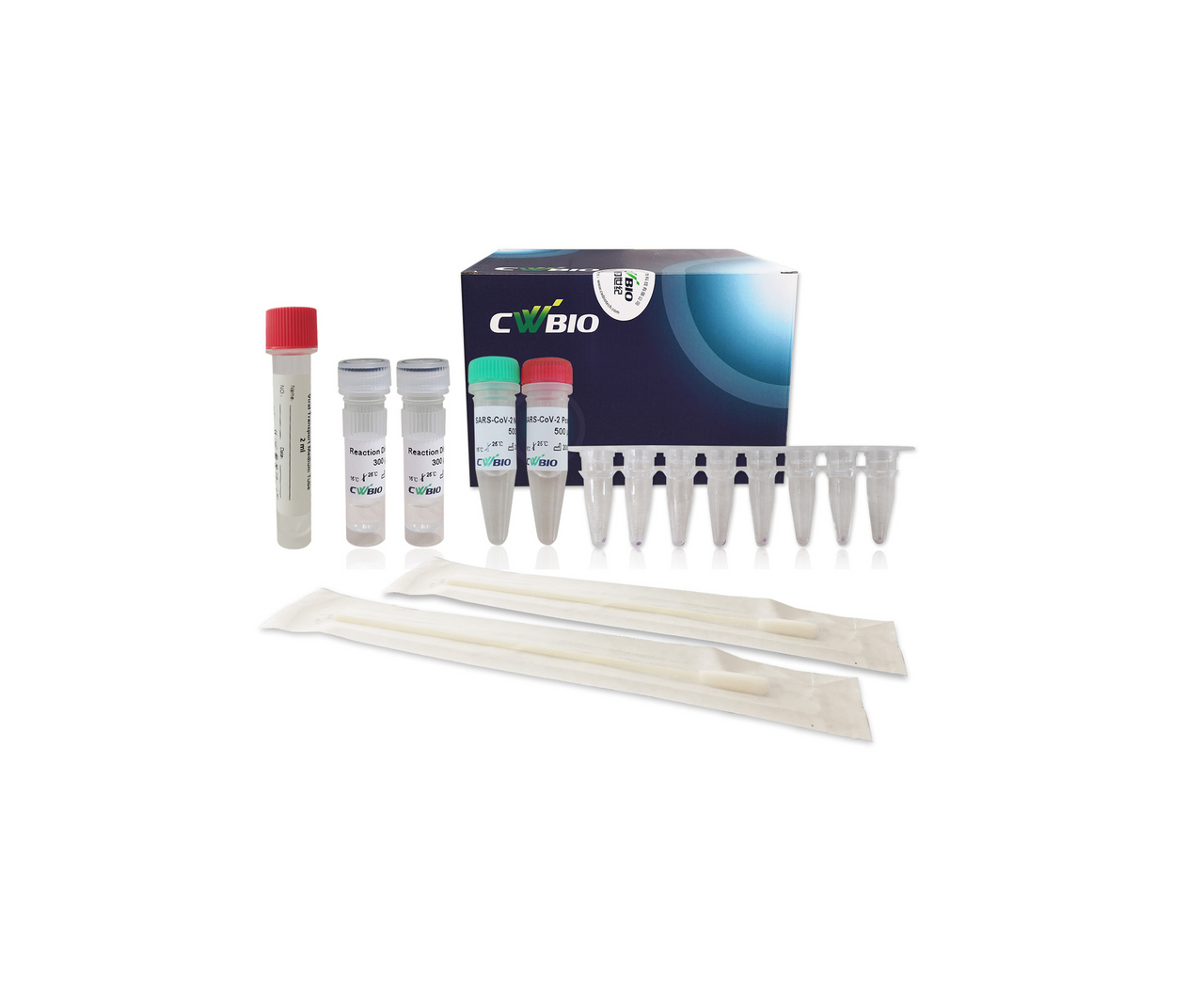
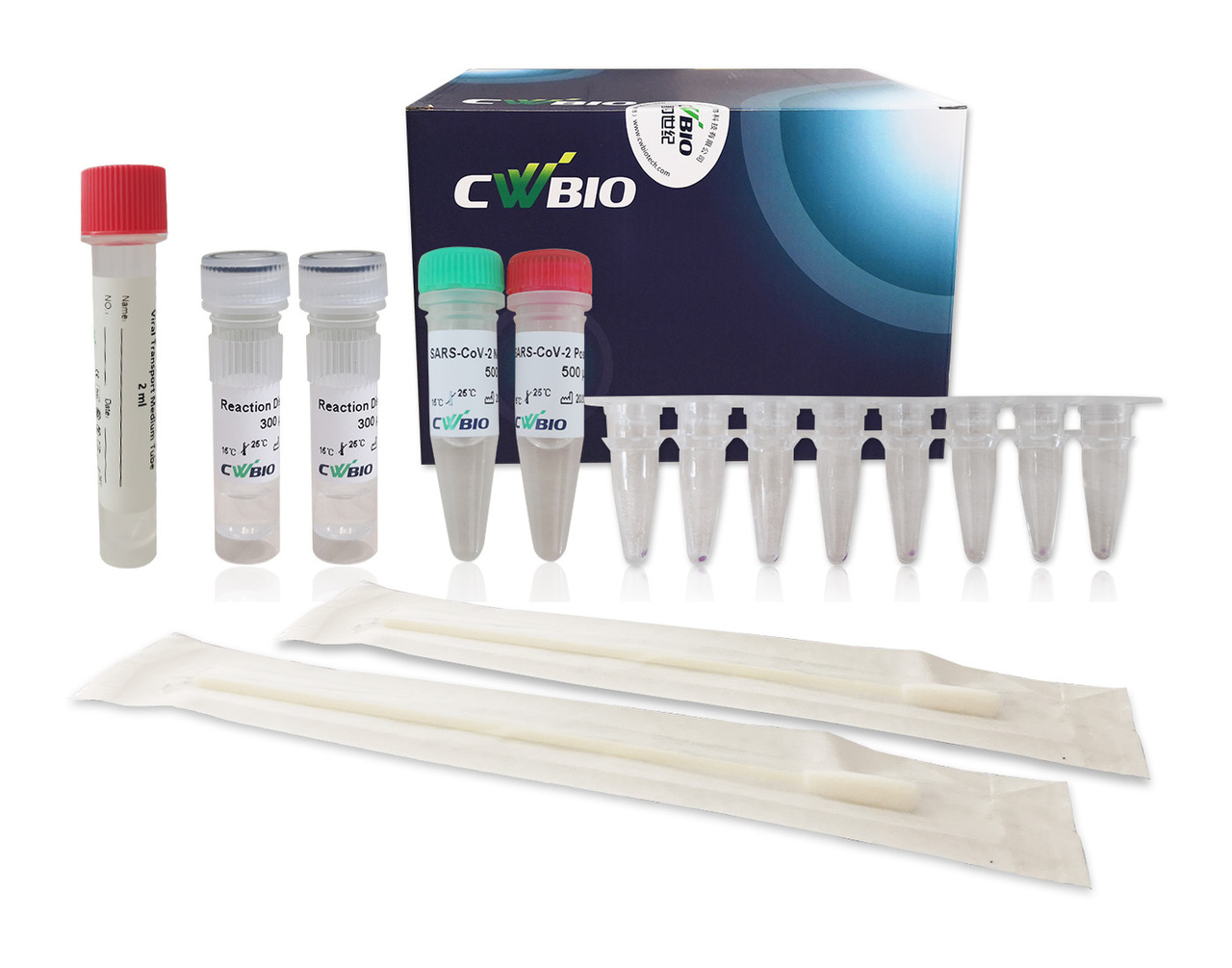
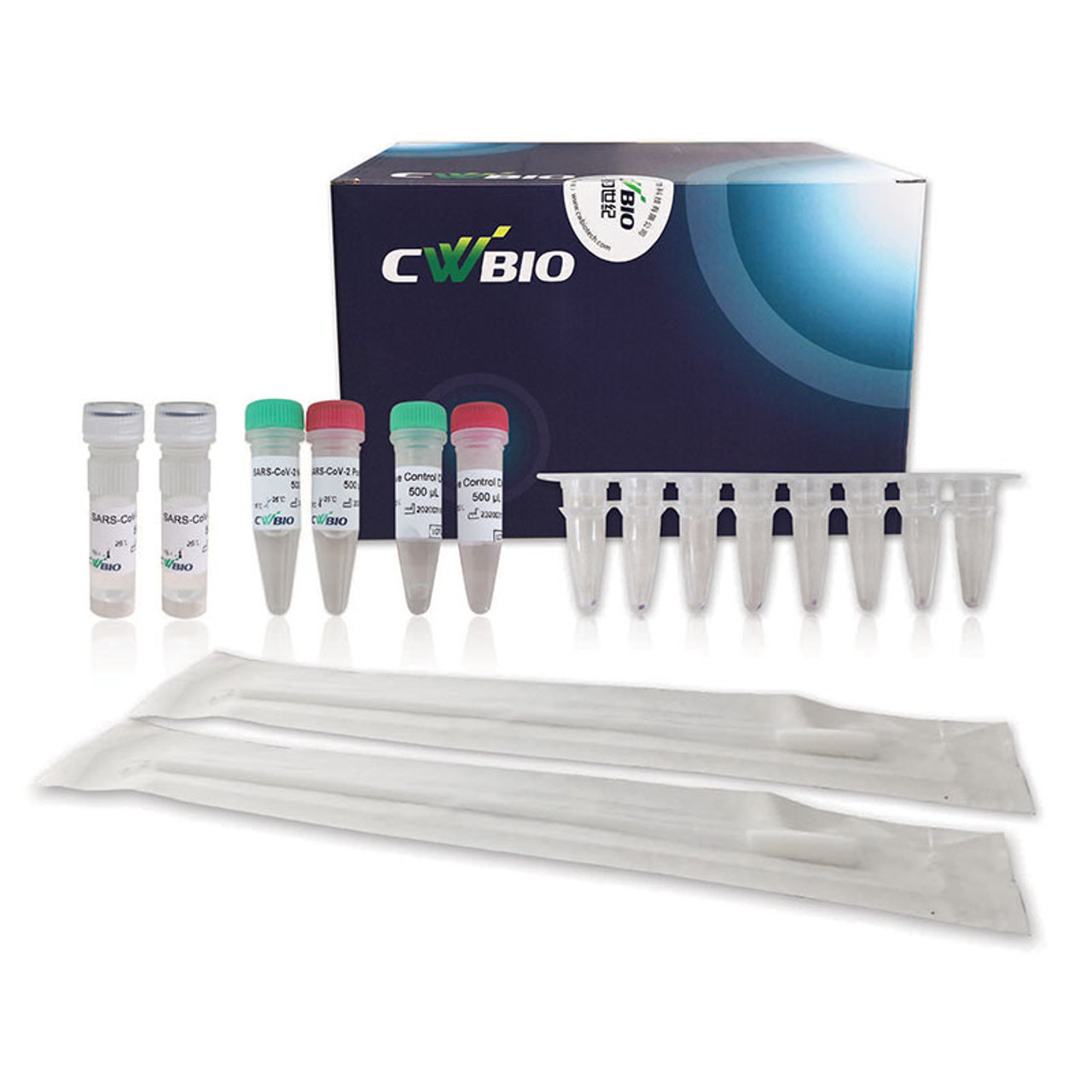
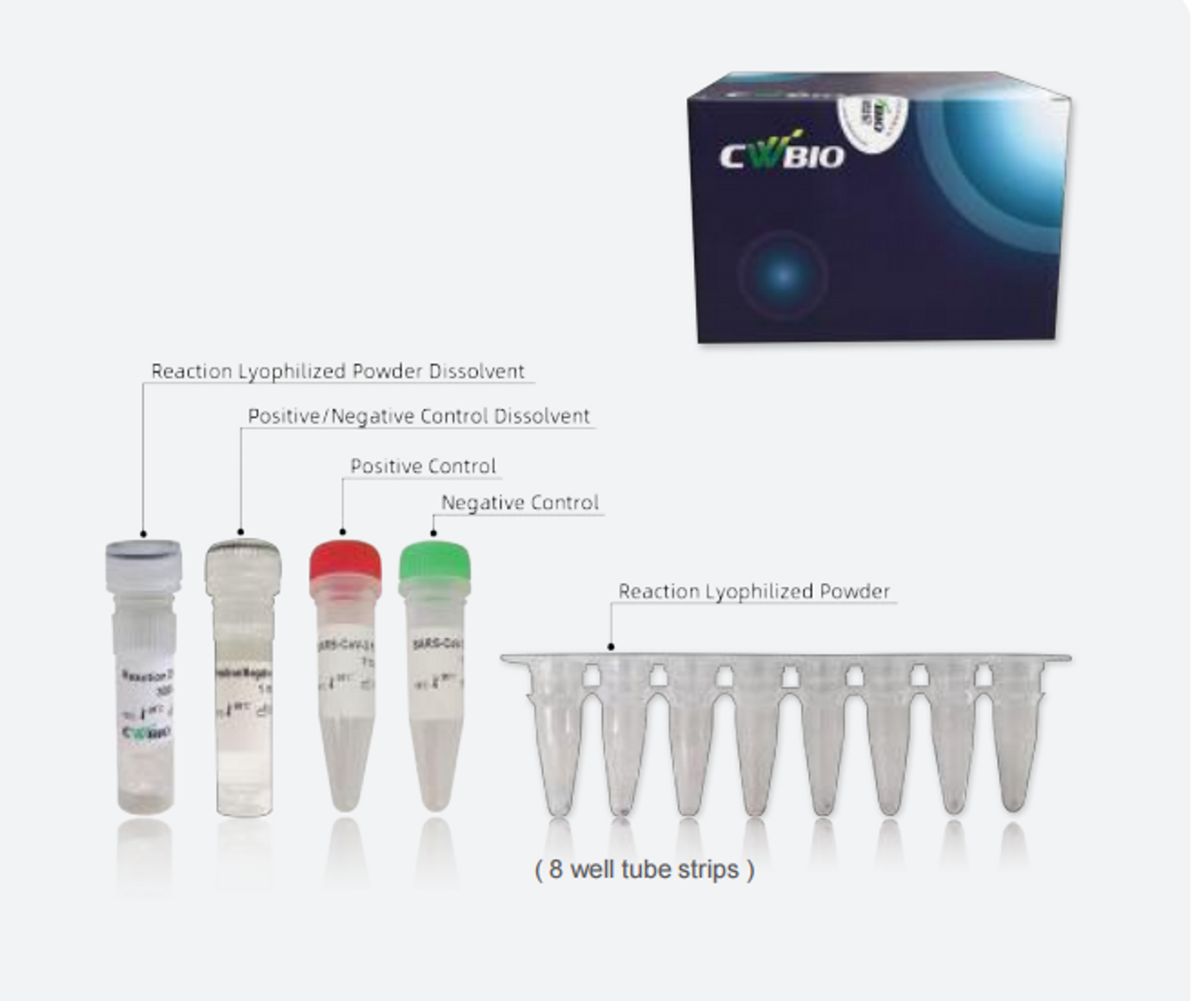
The Novel Coronavirus (SARS-CoV-2) Fast Nucleic Acid Detection Kit (PCR- Fluorescence Probing) is a real-time reverse transcription-polymerase chain reaction (rRT-PCR) test. The SARS-CoV-2 primer and probe set(s) is designed to detect specific ORF1ab and N genes from SARS-CoV-2 in oropharyngeal swabs from patients suspected of COVID-19 by their healthcare provider.
Request Quotation: us@genprice.com
CWBio FDA EUA SARS-CoV-2 Fast PCR Fluorescence Probing Kit DataSheet
The Novel Coronavirus (SARS-CoV-2) Fast Nucleic Acid Detection Kit (PCR- Fluorescence Probing) requires collection of oropharyngeal specimens with CoWin swabs followed by immediate placement in a special preservation solution (SARS-CoV-2 Collection Fluid containing Tris buffer, EDTA, NaOH, Tween 20, etc.) that allows testing to be performed without the isolation and purification of nucleic acids from specimens.
The Novel Coronavirus (SARS-CoV-2) Fast Nucleic Acid Detection Kit (PCR-Fluorescence Probing) is a real-time RT-PCR test intended for the qualitative detection of nucleic acid from SARS-CoV-2 in oropharyngeal swab specimens collected from individuals suspected of COVID-19 by their healthcare provider. Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C. §263a, that meet requirements to perform high complexity tests.
Results are for the identification of SARS-CoV-2 RNA. The SARS-CoV-2 RNA is generally detectable in respiratory specimens during the acute phase of infection. Positive results are indicative of the presence of SARS-CoV-2 RNA; clinical correlation with patient history and other diagnostic information is necessary to determine patient infection status. Positive results do not rule out bacterial infection or co-infection with other viruses. The agent detected may not be the definite cause of disease. Laboratories within the United States and its territories are required to report all results to the appropriate public health authorities.
Negative results do not preclude SARS-CoV-2 infection and should not be used as the sole basis for patient management decisions. Negative results must be combined with clinical observations, patient history, and epidemiological information.
The Novel Coronavirus (SARS-CoV-2) Fast Nucleic Acid Detection Kit (PCR-Fluorescence Probing) is intended for use by qualified clinical laboratory personnel specifically instructed and trained in the techniques of real-time PCR assays and in vitro diagnostic procedures. The Novel Coronavirus (SARS-CoV-2) Fast Nucleic Acid Detection Kit (PCR-Fluorescence Probing) is only for use under the Food and Drug Administration’s Emergency Use Authorization.
Specimen Collection and Storage Collection Process
1. Clean hands before sampling, tear open the package of Swabs, and take out the swab. Be careful not to touch the swab head.
2. Wipe the bilateral pharyngeal tonsils and the posterior pharyngeal wall with a swab over the base of the tongue.
3. After sampling, put the swab into a SARS-CoV-2 Collection Fluid tube to avoid contact with other parts.
4. Break the swab tip, discard stick and cap the tube containing samples.
Specimen Storage and Transportation
After sampling, swab samples need to be immediately placed in the tube with the SARS- CoV-2 Collection Fluid provided in this kit Swab samples treated with the SARS-CoV-2 Collection Fluid can be stored at 2-8 ℃ or - 80 ℃ for 10 days and can be transported at 2-8 ℃ for no more than 3 days.
Request Quotation: us@genprice.com
 Euro
Euro
 USD
USD
 British Pound
British Pound
 NULL
NULL