Product Description
HERPUD1 Antibody | 22-659 | ProSci
Host: Rabbit
Reactivity: Human, Mouse, Rat
Homology: N/A
Immunogen: Recombinant fusion protein containing a sequence corresponding to amino acids 1-240 of human HERPUD1 (NP_055500.1) .
Research Area: Cell Cycle, Signal Transduction
Tested Application: WB
Application: WB: 1:500 - 1:2000
Specificiy: N/A
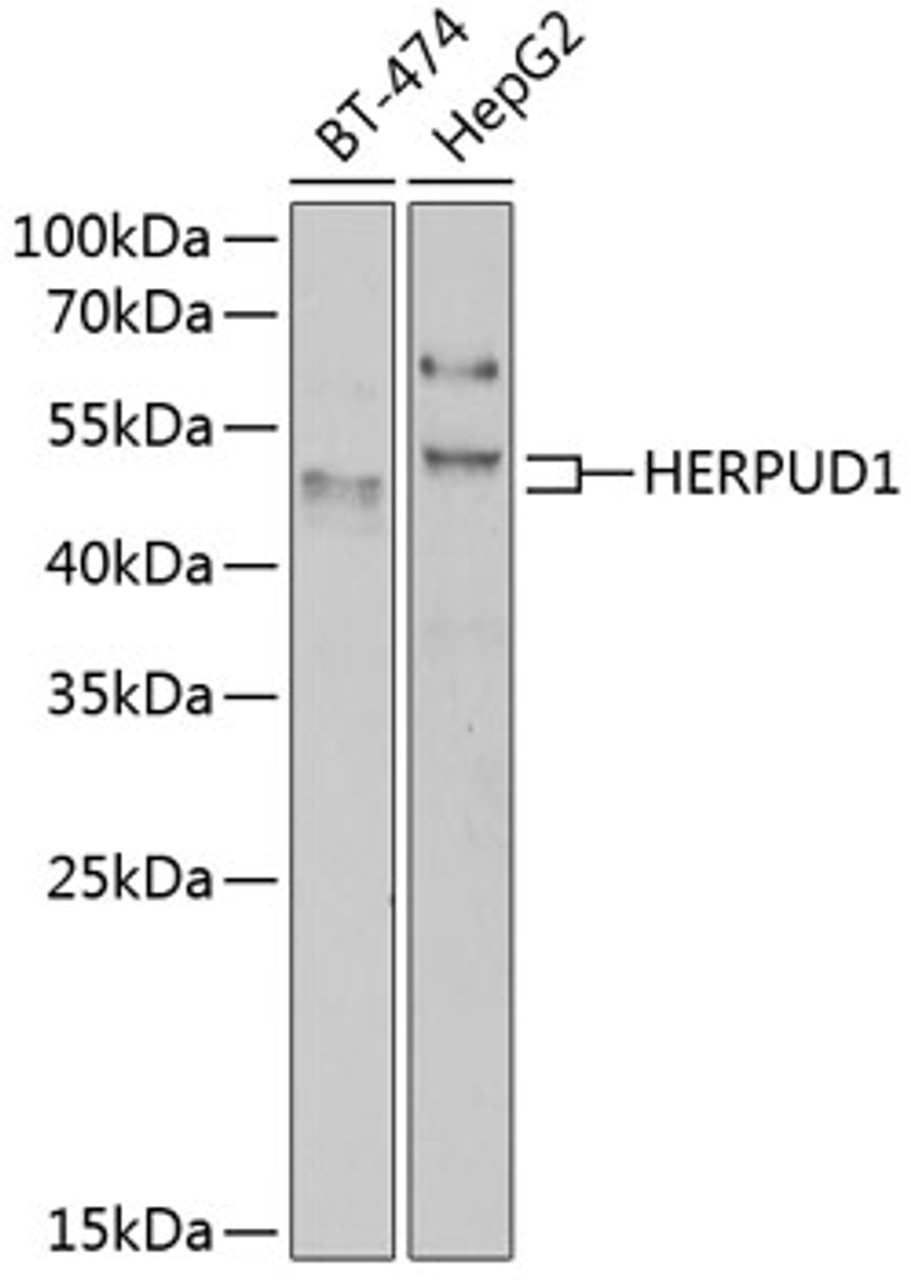
Positive Control 1: BT-474
Positive Control 2: HepG2
Positive Control 3: N/A
Positive Control 4: N/A
Positive Control 5: N/A
Positive Control 6: N/A
Molecular Weight: Observed: 44kDa
Validation: N/A
Isoform: N/A
Purification: Affinity purification
Clonality: Polyclonal
Clone: N/A
Isotype: IgG
Conjugate: Unconjugated
Physical State: Liquid
Buffer: PBS with 0.02% sodium azide, 50% glycerol, pH7.3.
Concentration: N/A
Storage Condition: Store at -20˚C. Avoid freeze / thaw cycles.
Alternate Name: Homocysteine-responsive endoplasmic reticulum-resident ubiquitin-like domain member 1 protein, Methyl methanesulfonate (MMF) -inducible fragment protein 1, HERPUD1, HERP, KIAA0025, MIF1
User Note: Optimal dilutions for each application to be determined by the researcher.
BACKGROUND: The accumulation of unfolded proteins in the endoplasmic reticulum (ER) triggers the ER stress response. This response includes the inhibition of translation to prevent further accumulation of unfolded proteins, the increased expression of proteins involved in polypeptide folding, known as the unfolded protein response (UPR) , and the destruction of misfolded proteins by the ER-associated protein degradation (ERAD) system. This gene may play a role in both UPR and ERAD. Its expression is induced by UPR and it has an ER stress response element in its promoter region while the encoded protein has an N-terminal ubiquitin-like domain which may interact with the ERAD system. This protein has been shown to interact with presenilin proteins and to increase the level of amyloid-beta protein following its overexpression. Alternative splicing of this gene produces multiple transcript variants encoding different isoforms. The full-length nature of all transcript variants has not been determined.
 Euro
Euro
 USD
USD
 British Pound
British Pound
 NULL
NULL