Product Description
FEN1 Antibody | 61-554 | ProSci
Host: Rabbit
Reactivity: Human
Homology: Predicted species reactivity based on immunogen sequence: Bovine
Immunogen: This FEN1 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 243-272 amino acids from the Central region of human FEN1.
Research Area: Other
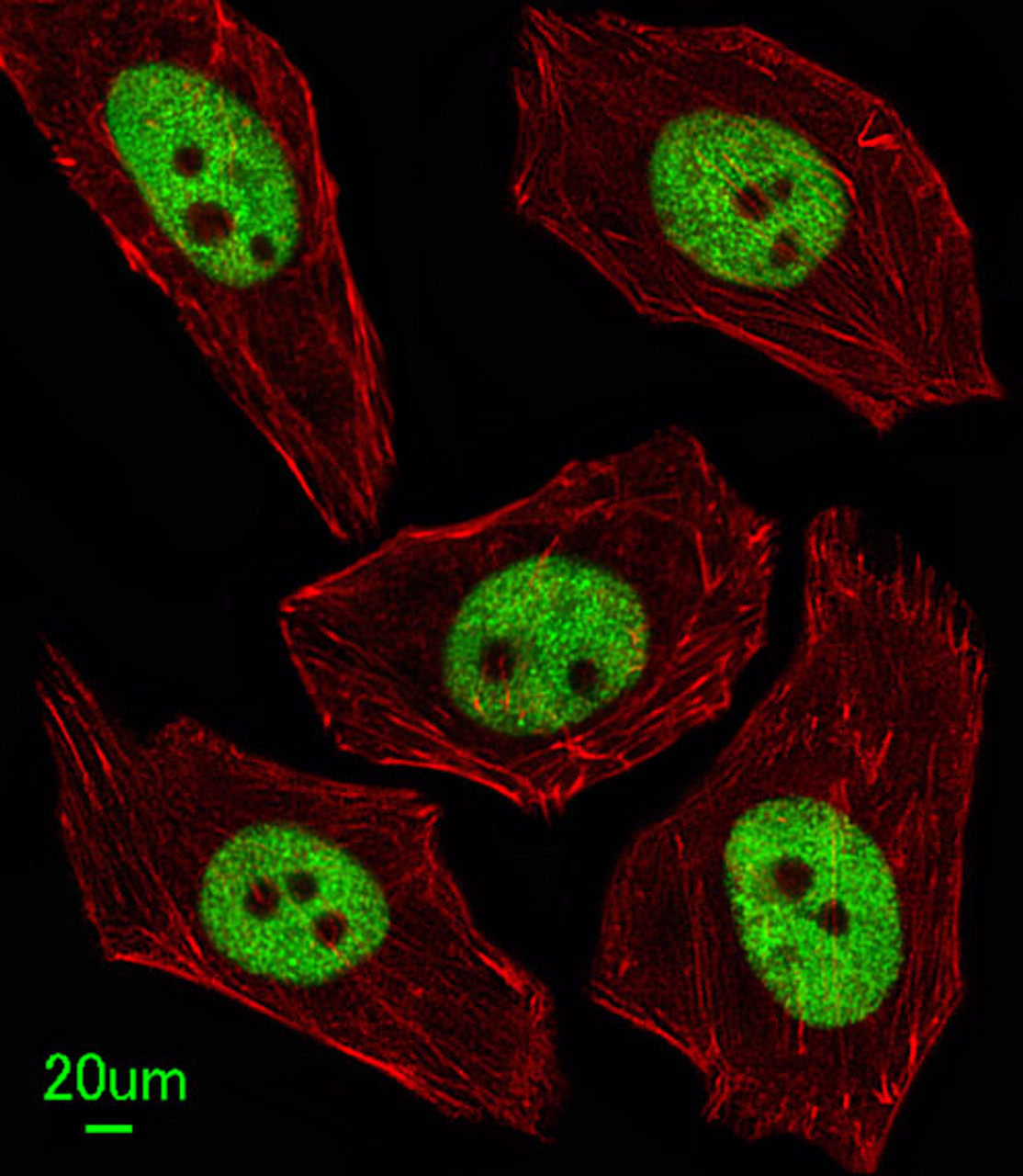
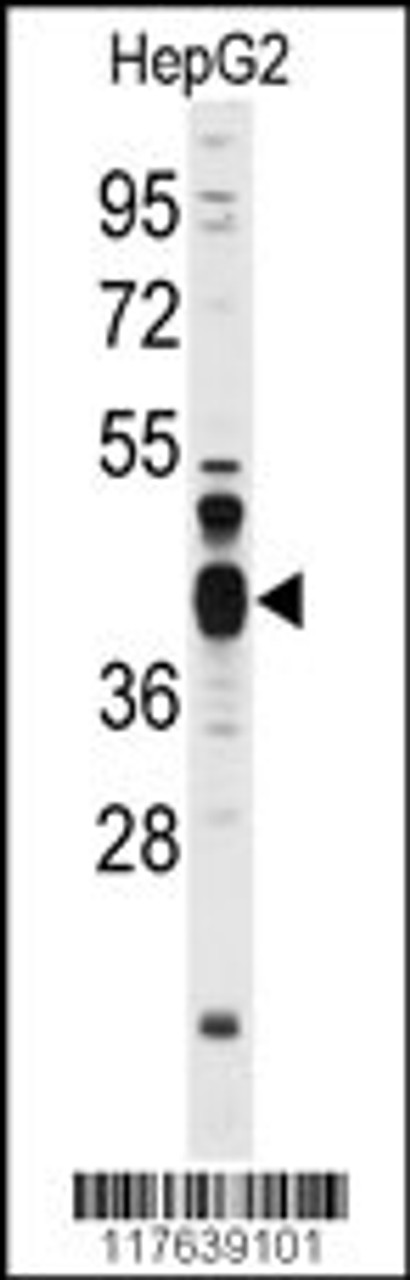
Tested Application: WB, IF
Application: For IF starting dilution is: 1:100
For WB starting dilution is: 1:1000
Specificiy: N/A
Positive Control 1: N/A
Positive Control 2: N/A
Positive Control 3: N/A
Positive Control 4: N/A
Positive Control 5: N/A
Positive Control 6: N/A
Molecular Weight: 43 kDa
Validation: N/A
Isoform: N/A
Purification: This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis
Clonality: Polyclonal
Clone: N/A
Isotype: Rabbit Ig
Conjugate: Unconjugated
Physical State: Liquid
Buffer: Supplied in PBS with 0.09% (W/V) sodium azide.
Concentration: batch dependent
Storage Condition: Store at 4˚C for three months and -20˚C, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures.
Alternate Name: Flap endonuclease 1 {ECO:0000255|HAMAP-Rule:MF_03140}, FEN-1 {ECO:0000255|HAMAP-Rule:MF_03140}, 31-- {ECO:0000255|HAMAP-Rule:MF_03140}, DNase IV, Flap structure-specific endonuclease 1 {ECO:0000255|HAMAP-Rule:MF_03140}, Maturation factor 1, MF1, hFEN-1, FEN1 {ECO:0000255|HAMAP-Rule:MF_03140}, RAD2
User Note: Optimal dilutions for each application to be determined by the researcher.
BACKGROUND: FEN1 removes 5' overhanging flaps in DNA repair and processes the 5' ends of Okazaki fragments in lagging strand DNA synthesis. Direct physical interaction between this protein and AP endonuclease 1 during long-patch base excision repair provides coordinated loading of the proteins onto the substrate, thus passing the substrate from one enzyme to another. This protein is a member of the XPG/RAD2 endonuclease family and is one of ten proteins essential for cell-free DNA replication. DNA secondary structure can inhibit flap processing at certain trinucleotide repeats in a length-dependent manner by concealing the 5' end of the flap that is necessary for both binding and cleavage by the protein encoded by this gene. Therefore, secondary structure can deter the protective function of this protein, leading to site-specific trinucleotide expansions.
 Euro
Euro
 USD
USD
 British Pound
British Pound
 NULL
NULL