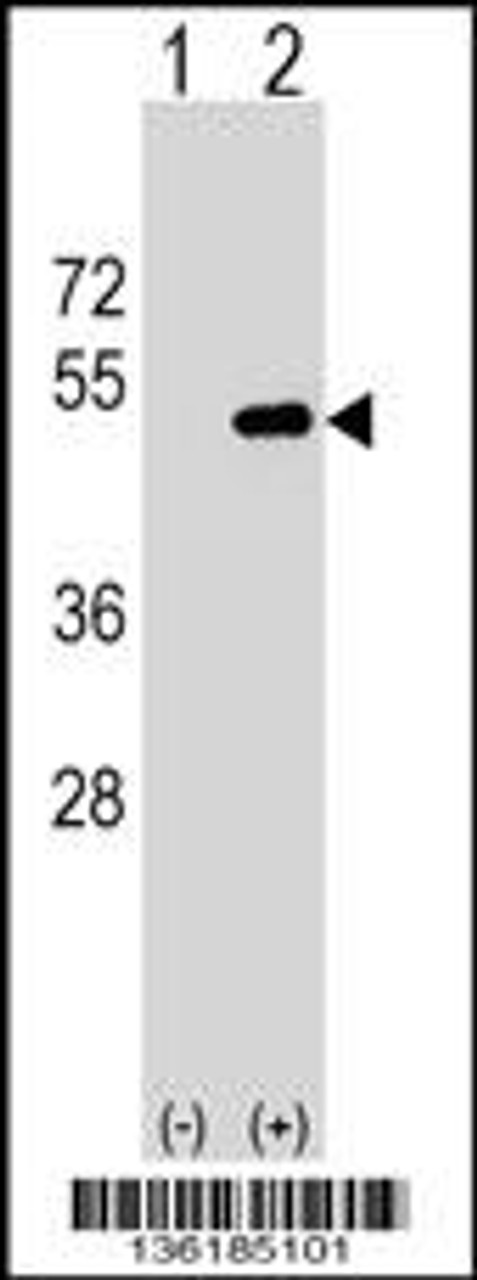
Product Description
FNTA Antibody | 59-614 | ProSci
Host: Rabbit
Reactivity: Human
Homology: Predicted species reactivity based on immunogen sequence: Mouse, Rat
Immunogen: This FNTA antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 51-79 amino acids from the N-terminal region of human FNTA.
Research Area: Cell Cycle, Signal Transduction
Tested Application: WB
Application: For WB starting dilution is: 1:1000
Specificiy: N/A
Positive Control 1: N/A
Positive Control 2: N/A
Positive Control 3: N/A
Positive Control 4: N/A
Positive Control 5: N/A
Positive Control 6: N/A
Molecular Weight: 44 kDa
Validation: N/A
Isoform: N/A
Purification: This antibody is purified through a protein A column, followed by peptide affinity purification.
Clonality: Polyclonal
Clone: N/A
Isotype: Rabbit Ig
Conjugate: Unconjugated
Physical State: Liquid
Buffer: Supplied in PBS with 0.09% (W/V) sodium azide.
Concentration: batch dependent
Storage Condition: Store at 4˚C for three months and -20˚C, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures.
Alternate Name: Protein farnesyltransferase/geranylgeranyltransferase type-1 subunit alpha, CAAX farnesyltransferase subunit alpha, FTase-alpha, Ras proteins prenyltransferase subunit alpha, Type I protein geranyl-geranyltransferase subunit alpha, GGTase-I-alpha, FNTA
User Note: Optimal dilutions for each application to be determined by the researcher.
BACKGROUND: Prenyltransferases can attach either a farnesyl group or a geranylgeranyl group in thioether linkage to the cysteine residue of proteins with a C-terminal CAAX box. CAAX geranylgeranyltransferase and CAAX farnesyltransferase are heterodimers that share the same alpha subunit but have different beta subunits. This gene encodes the alpha subunit of these transferases. Alternative splicing results in multiple transcript variants. Related pseudogenes have been identified on chromosomes 11 and 13.
 Euro
Euro
 USD
USD
 British Pound
British Pound
 NULL
NULL